Scientists lay out plan to use CO2 emissions for good, when we can capture them
While scientists develop ways to affordably capture CO2 waste and convert it into useful molecules for industry, a team of researchers has outlined a vision for what can be made with the byproducts and how to go about it.
In a study published Tuesday in the journal Joule, the team identifies a series of small molecules that could benefit industry and could be produced by converting captured CO2.
For energy storage demands, ethane, hydrogen and methane could be used in biofuels while ethylene and ethanol could serve as the building blocks for an array of consumer products. CO2-derived formic acid, on the other hand, could be used by the pharmaceutical industry or as fuel in fuel cells.
“Similar to how a plant takes carbon dioxide, sunlight, and water to make sugars for itself, we are interested in using technology to take energy from the sun or other renewable sources to convert CO2 into small building block molecules which can then be upgraded using traditional means of chemistry for commercial use,” said co-first author Phil De Luna, a doctoral candidate at the University of Toronto.
“We’re taking inspiration from nature and doing it faster and more efficiently.”
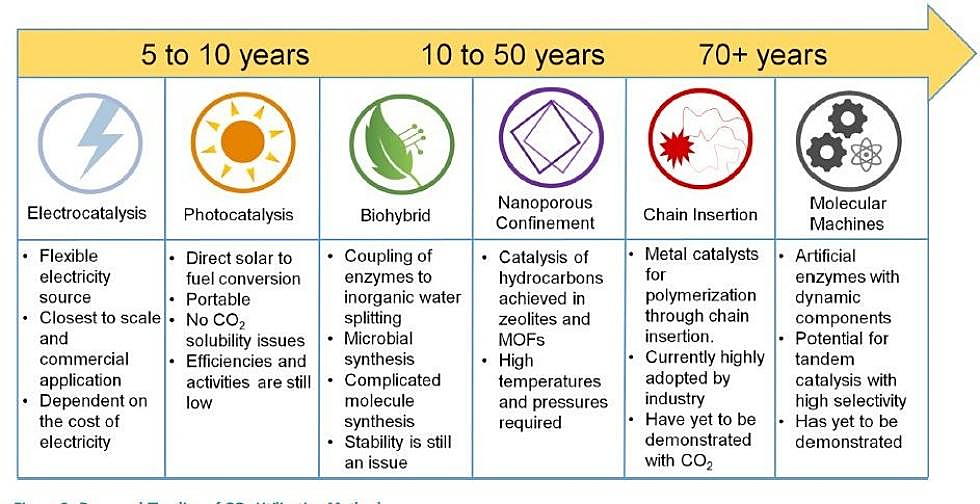
Existing technologies that can capture CO2 waste are still fairly new and startups currently working on strategies for commercial use. With these factors in mind, the team predicts that significant improvements to make CO2 capture and conversion a reality will occur over the next few decades.
Within five to 10 years, electrocatalysis – which stimulates chemical reactions through electricity – could be a viable avenue into the process, while molecular machines or nanotechnology could drive conversion 50 years or longer down the line.
“This is still technology for the future,” said co-first author Oleksandr Bushuyev, a postdoctoral fellow at the University of Toronto, “but it’s theoretically possible and feasible, and we’re excited about its scale-up and implementation.
“If we continue to work at this, it’s a matter of time before we have power plants where CO2 is emitted, captured, and converted.”
The team does note limitations to carbon capture and conversion. First, the process has been criticized as being too expensive, particularly due to the cost of electricity used to make these chemical reactions occur. However, these costs will likely be reduced as renewable energy becomes widespread. Second, only a few factories with significant carbon footprints emit pure CO2, which is needed for conversion, but technology that could help with this problem is being developed.
“The motivation to write this piece is that we wanted clear insight into whether this could be economically viable, and whether it’s worth the time to invest in it,” said De Luna. “This paper images a pathway for what we can do with carbon dioxide conversion in the coming decades.”
The research was financially supported by the CIFAR Bio-Inspired Solar Energy program, the Natural Sciences and Engineering Research Council of Canada, and the Ontario Research Fund.
